Modulation of protein release from penta-block copolymer microspheres European Journal of Pharmaceutics and Biopharmaceutics 152, 175–182 (2020)
European Journal of Pharmaceutics and Biopharmaceutics 152, 175–182 (2020).
Minh-Quan Le, Jean-Christophe Gimel, Xavier Garric, Thao-Quyen Nguyen-Pham, Cédric Paniagua, Jérémie Riou, Marie-Claire Venier-Julienne,
ABSTRACT
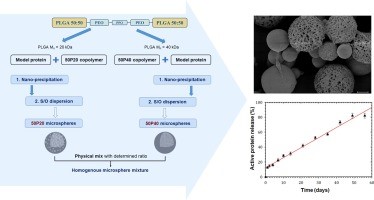
Releasing a protein according to a zero-order profile without protein denaturation during the polymeric microparticle degradation process is very challenging. The aim of the current study was to develop protein-loaded microspheres with new PLGA based penta-block copolymers for a linear sustained protein release. Lysozyme was chosen as model protein and 40 µm microspheres were prepared using the solid-in-oil-in-water solvent extraction/evaporation process. Two types of PLGA-P188-PLGA penta-block copolymers were synthetized with two PLGA-segments molecular weight (20 kDa or 40 kDa). The resulting microspheres (50P20-MS and 50P40-MS) had the same size, an encapsulation efficiency around 50–60% but different porosities. Their protein release profiles were complementary: linear but non complete for 50P40-MS, non linear but complete for 50P20-MS. Two strategies, polymer blending and microsphere mixing, were considered to match the release to the desired profile. The (1:1) microsphere mixture was successful. It induced a bi-phasic release with a moderate initial burst (around 13%) followed by a nearly complete linear release for 8 weeks. This study highlighted the potential of this penta-block polymer where the PEO block mass ratio influence clearly the Tg and consequently the microsphere structure and the release behavior at 37 °C. The (1:1) mixture was a starting point but could be finely tuned to control the protein release.